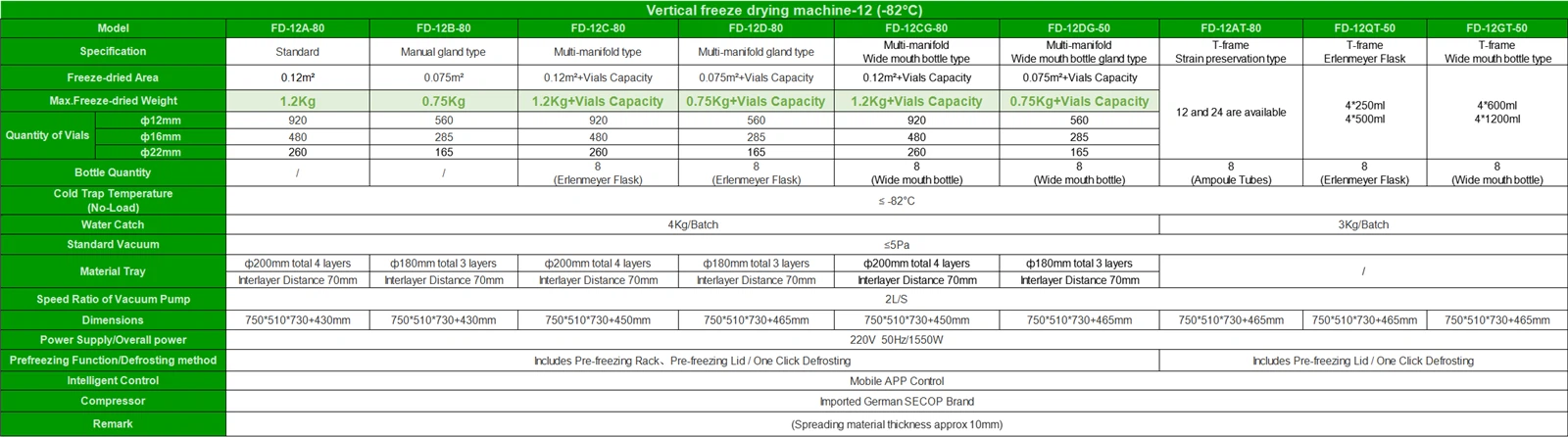
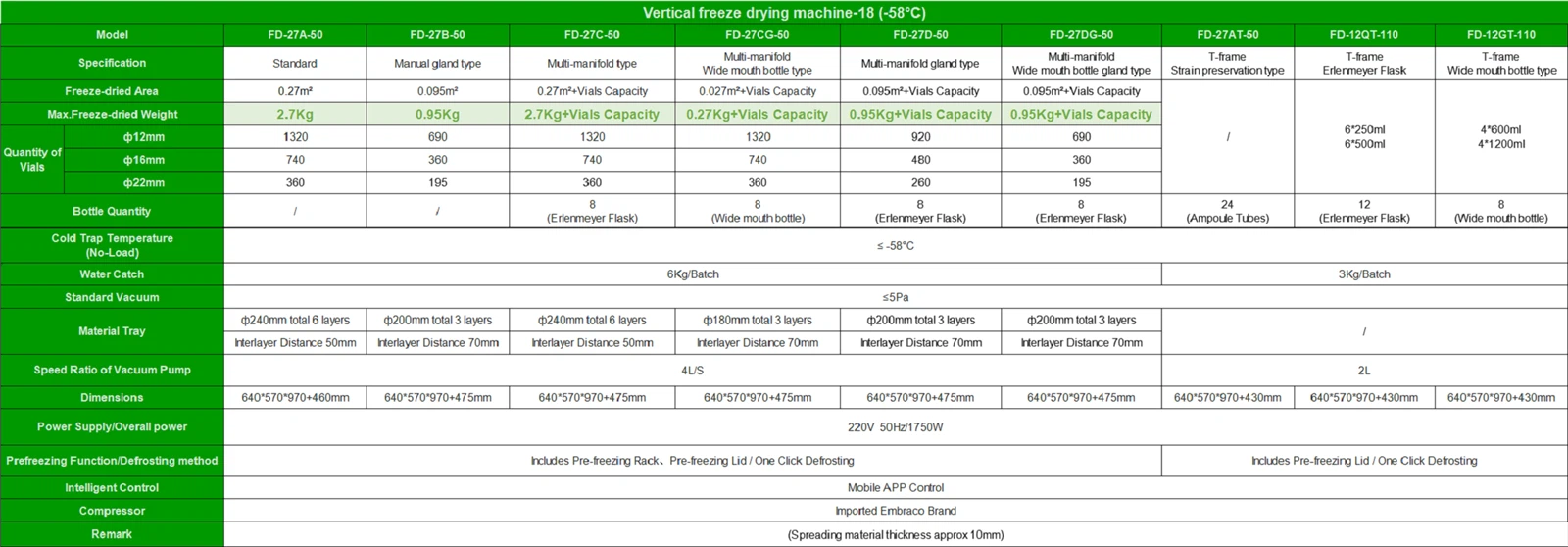
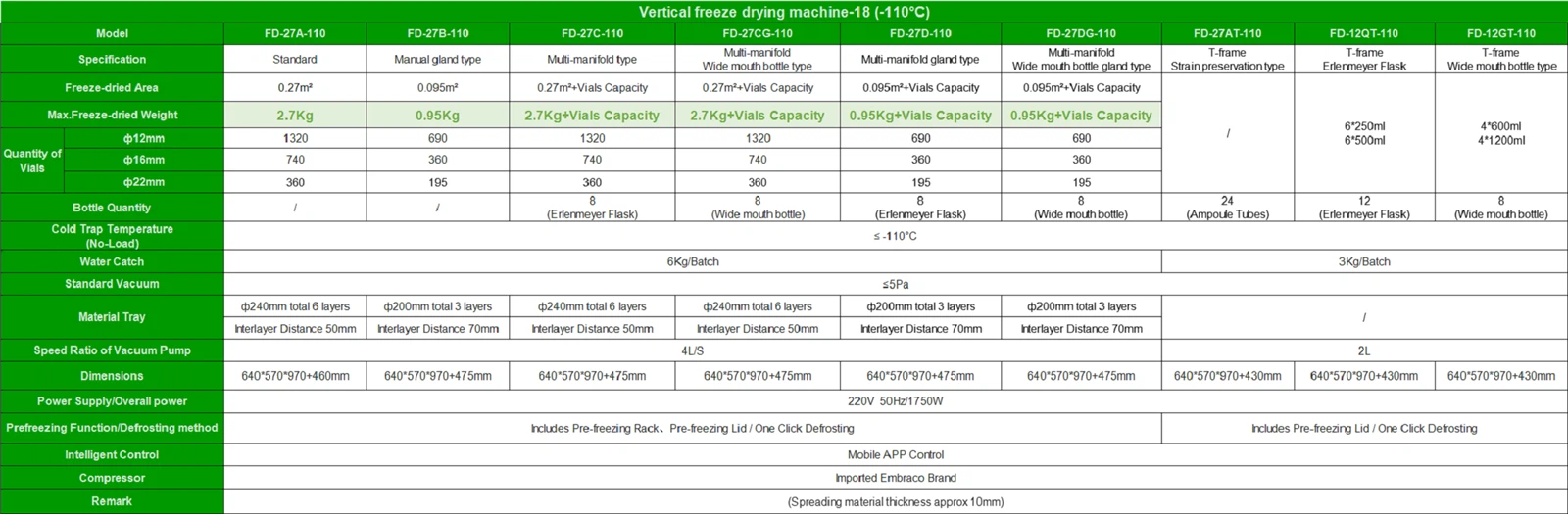
GMP certification




Drug GMP certification is a system in which the country implements drug GMP supervision and inspection on drug production enterprises (workshops) and drug varieties in accordance with the law and obtains recognition. It is a scientific and advanced management method to ensure the stability, effectiveness, and safety of drug quality. The soul of GMP is to ensure the quality of drugs. Passing GMP certification is only the minimum standard for pharmaceutical production enterprises, not the final result, nor is it a lifelong guarantee. The key to GMP certification lies in how to implement GMP in practice, and implement various
GMP management systems without any discount and conscientiously. The company first transforms the certification team into a working group as a permanent organization responsible for annual training and regular inspections of implementation work; The second is to establish a quality reward system and a quality salary system, linking the implementation of GMP and the first pass rate of products to worker wages, and linking them to bonuses for management and technical personnel positions; The third is to establish a dedicated GMP supervisor in each workshop, responsible for inspecting and supervising the entire production process, and formulating early, fast, and strict work requirements. Early detection of problems results in minimal losses, fast inspection, high production efficiency, and strict supervision with minimal errors; The fourth is to formulate standardized rules for disciplinary actions, requiring them to be both strict in quality and public; The fifth is to strictly control the entry of raw and auxiliary packaging materials, the supervision of the production process, and the release of finished products from the factory to ensure that 100% of the products are qualified; Six is to strengthen the quality awareness and GMP awareness of employees, and regularly recite the "GMP three character song" and so on.
Application in chemical laboratories for the lyophilization of peptides
► Basic principle of peptide lyophilization
- The working principle of peptide lyophilizer is mainly based on two processes: low temperature condensation and sublimation drying. In the low-temperature condensation process, the peptide solution is rapidly cooled, and the water in it forms ice crystals at low temperature. Subsequently, in the sublimation drying process, the ice crystals are directly transformed from the solid state to the gaseous state under the vacuum condition, so as to remove the water and realize the drying of peptide drugs.
► The advantages of desktop freeze dryer
Activity protection
Vacuum freeze-drying technology can be carried out in a low-temperature, low-oxygen environment, which effectively reduces the risk of degradation of biomolecules in a high-temperature or oxidizing environment, and greatly preserves the activity and function of peptides and other biological products.
Easy to re-solubilize
Lyophilized products have a porous structure and can be re-solubilized quickly in a short time to restore the original solution state, which is convenient for subsequent experiments or clinical applications.
Simplified transportation and storage
lyophilization can also reduce the volume of peptide, which is convenient for transportation and storage.
► Application of desktop lyophilizer in peptide lyophilization
- Preparation of insoluble peptides: Through lyophilization, peptides can be prepared into powder form to improve their solubility and bioavailability.
- Preparation of long-lasting peptides: By controlling the parameters in the freeze-drying process, such as temperature, pressure and time, the crystalline form and pore size of peptides can be changed, thus realizing the long-lasting and slow release of drugs.
- R&D and production of peptide drugs: It plays an important role in the R&D and production of peptide drugs, providing strong support for preclinical research and commercial production of drugs.
Peptides dried by the Benchtop Lyophilizer
► The basic role of polypeptide
- Regulate physiological functions: peptides can regulate signaling inside and outside the cell by binding with specific receptors, thus affecting the physiological activities of the body.
- Promote cell growth: some peptides have the ability to promote cell proliferation and differentiation, and have a positive effect on tissue repair and regeneration.
- Enhance immune response: peptides can be used as immunomodulators to enhance the body's immune response and improve resistance to pathogens.
- Improve nutritional absorption: peptides can be used as nutritional supplements to help improve protein absorption and utilization, which is especially important for specific groups such as the elderly and athletes.
► The protective effect of vacuum freeze-drying technology on peptides
- Activity protection: Vacuum freeze-drying technology can be carried out in a low-temperature, low-oxygen environment, effectively reducing the risk of degradation of biomolecules in a high-temperature or oxidizing environment, and greatly preserving the activity and function of peptides.
- Structural integrity: The sublimation process avoids the damage to the structure of the sample caused by the melting of ice crystals, and maintains the original morphology and microstructure of the sample, which helps the peptide to maintain its original biological activity.
- Long-term storage: the dried peptide has extremely low water content, which effectively inhibits the growth of microorganisms and the occurrence of chemical reactions, significantly extends the shelf life of the product, and facilitates long-distance transportation and long-term storage.
► Application of dried peptide in specific fields
- Biomedical field: peptide drugs have attracted much attention because of their high activity and specificity. Peptide drugs dried by vacuum freeze-drying technology can maximize the retention of its activity and extend the shelf life, providing strong support for preclinical research and commercial production of drugs.
- Cosmetic field: Peptides are commonly used as anti-aging ingredients in cosmetics, such as promoting collagen and elastin production, reducing inflammation, and locking in skin moisture. Vacuum freeze-dried peptides are able to maintain their activity better, thus exerting anti-aging effects more effectively.
In summary, benchtop lyophilizer has significant advantages and wide application prospects for the lyophilization of peptides in chemical laboratories. Through reasonable selection and use, it can provide powerful technical support for the R&D and production of peptide drugs.
What should be done if a sample leak occurs
Emergency treatment
Shut down and power off
Stop the freeze-dryer operation immediately and cut off the power supply to prevent further leakage or other safety issues.
Isolated leak area
Quickly isolate the leak area to prevent the spill from spreading to a wider area.
Use appropriate protective equipment, such as gloves, masks, and goggles, to prevent direct contact with leaks or inhalation of harmful gases.
Leakage collection
Use appropriate containers or tools to collect leaked samples and prevent them from flowing into the drainage system or mixing with other substances.
If the leakage is liquid, absorbent materials (such as gauze, paper towels, etc.) can be used to absorb; If the spill is solid or powder, it can be collected using a shovel or brush.
Inspection and evaluation

Check the cause of the leak
Carefully examine the leak site to determine whether the leak is due to equipment failure, improper operation, or a problem with the sample itself.
Check whether the seals, valves, and pipes of the device are damaged or loose.
Assess the extent of the leak
Assess the severity of the spill based on the quantity, nature and spread of the spill.
If the leak is serious, it may be necessary to immediately notify the relevant departments or professionals for emergency treatment.

Cleaning and disinfection

Clean the leak area
Thoroughly clean the spill area with an appropriate detergent or disinfectant to remove residue.
Pay attention to personal protection during cleaning to avoid direct contact with leaks.
Disinfection treatment
Disinfect the cleaned area to prevent the growth of microorganisms such as bacteria or viruses.
Relevant disinfection codes and standards should be followed during the disinfection process.

Follow-up treatment
Repair or replace equipment
If the leak is caused by equipment failure, the relevant parts should be repaired or replaced as soon as possible.
After repair or replacement, a thorough inspection and test should be carried out to ensure that the equipment can operate properly and no leakage will occur.
Records and reports
Record information such as the time, location, cause, and treatment process of the leakage event.
Report the record to the relevant department or responsible person for subsequent analysis and improvement.
Training and Publicity
Conduct relevant training and publicity for operators to improve their safety awareness and operation skills.
Emphasize the importance of equipment maintenance and maintenance to ensure that the equipment is in good working condition.
A benchtop lyophilizer, or freeze dryer, is a laboratory-scale device engineered to remove moisture from biological, pharmaceutical, or food samples through sublimation, ensuring long-term stability without compromising quality. Its compact design and functionality make it a preferred choice for research labs, pilot manufacturing, and applications where space and budget constraints are critical.
The process involves three stages: freezing the sample to solidify water, applying a vacuum to lower pressure, and gently heating to sublimate ice directly into vapor. A cold trap condenses the vapor, preventing reabsorption. Benchtop models typically offer temperature ranges of −50 °C to +60 °C and vacuum levels of 0.1–1 mbar, tailored for delicate materials.